On September 21, a research paper entitled “Enantioselective [2+2]-cycloadditions with triplet photoenzymes” was published in Nature by the group of Prof. Zhong Fangrui and Wu Yuzhou from School of Chemistry and Chemical Engineering, Huazhong University of Science and Technology (HUST), and Prof. Chen Xi from Northwestern University was published in Nature.


Naturally evolved enzymes, despite astonishingly large variety and function diversity, operate predominantly through thermochemical activation. Integrating prominent photocatalysis modes into proteins such as triplet energy transfer could create artificial photoenzymes that expand the scope of natural biocatalysis. The Wu and Zhong group had exploited genetically reprogrammed, chemically evolved photoenzymes embedded with a synthetic triplet photosensitizer that are capable of excited state enantioinduction. Structural optimization through four rounds of directed evolution afforded proficient variants for the enantioselective intramolecular [2+2] photocycloaddition of indole derivatives with good substrate generality and excellent enantioselectivities (up to 99% enantiomeric excess). A crystal structure of the photoenzyme-substrate complex elucidated the noncovalent interactions which mediate reaction stereochemistry. This study expands the energy transfer reactivity of artificial triplet photoenzymes in a supramolecular protein cavity and unlocks an integrated approach to valuable enantioselective photochemical synthesis that is not accessible with either the synthetic or the biological world alone.
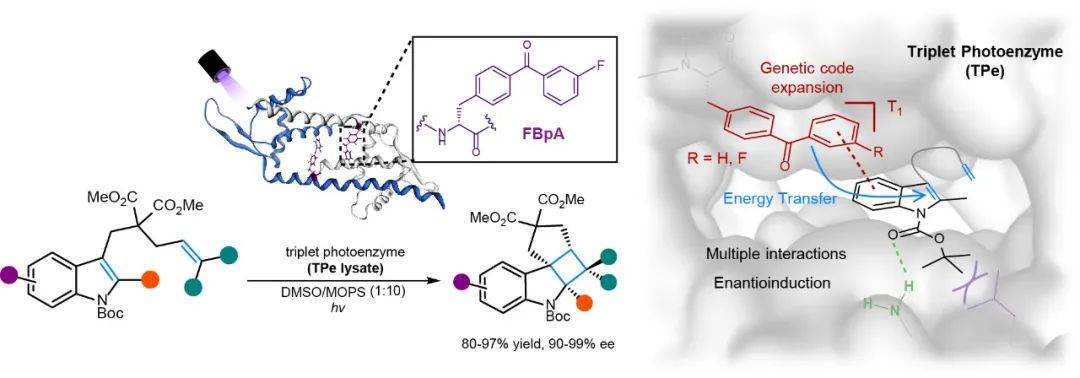
The [2+2] photocycloaddition catalyzed by triplet photoenzyme (TPe).
Illustration of the "Triplet Photoenzyme"
They created an entirely new photocatalytic center in TPe. Rounds of directed evolution based on a focused library identified evolved photoenzyme variants that enabled enantioselective intramolecular [2+2] photocycloadditions of indole derivatives (15 entries, 80-97% yields, all ≥ 90% e.e.) under aerobic conditions. This study demonstrates that the merger of versatile synthetic photocatalysts with a macromolecular protein can impart new reactivity paradigm that fundamentally expands the catalytic repertoire of enzymes for mediating new-to-nature photochemical reactions. The intricate and delicate protein cavity provides a superlative chiral environment for controlling the notoriously difficult enantiotopic selectivity of excited state bonding. Given the power of computation-aided artificial enzyme design and the continuous expansion of reprogrammed genetic codes for incorporating more robust noncanonical amino acid photocatalysts, triplet photoenzymes have the promise to become a general solution for various valuable enantioselective photochemical transformations.
The work was accomplished by the research group of Prof. Wu Yuzhou and Prof. Zhong Fangrui in collaboration with Prof. Chen Xi, Associate Prof. Zhao Weining of Shenzhen University of Technology, and Prof. Liao Rongzhen of Huazhong University of Science and Technology. They provided support in protein crystal analysis, protein mass spectrometry, and theoretical calculation, respectively. This work was funded by the National Key R&D Program for Synthetic Biology, the National Natural Science Foundation of China and other projects, and was supported by Analytical and Testing Centre of HUST, Analytical and Testing Centre of School of Chemistry and Chemical Engineering (HUST) and Research Core Facilities for Life Science (HUST) for instruments, as well as the staff from BL18U1 beamline of National Facility for Protein Science in Shanghai (NFPS) at Shanghai Synchrotron Radiation Facility, for assistance in data collection.
This paper is a breakthrough achievement of the research group led by Prof. Wu Yuzhou and Prof. Zhong Fangrui in the intersection field of chemical biology and organic synthetic chemistry. Prof. Wu Yuzhou has long been engaged in the chemical modification of proteins, nucleic acids and other natural biological macromolecules and research in the field of synthetic biology. She has presided over the National Key R&D Program "Synthetic Biology" key special project, the National Natural Science Foundation of China, and the Hubei Provincial Key R&D Program. Her research interests include the development and application of non-natural enzymes and protein materials, chemical modification of nucleic acids and nanomedicine materials. She has published more than 80 papers in international leading journals including Nature, Chemical Review, JACS, and Angew Chem. Prof. Zhong Fangrui has been engaged in research in the field of asymmetric catalysis and biocatalysis. He has published more than 50 papers in journals including Nature, JACS, and Angew Chem.
Link to the paper: https://www.nature.com/articles/s41586-022-05342-4
Link to the research group: http://faculty.hust.edu.cn/zhongfangrui/zh_CN/index.htm
